|
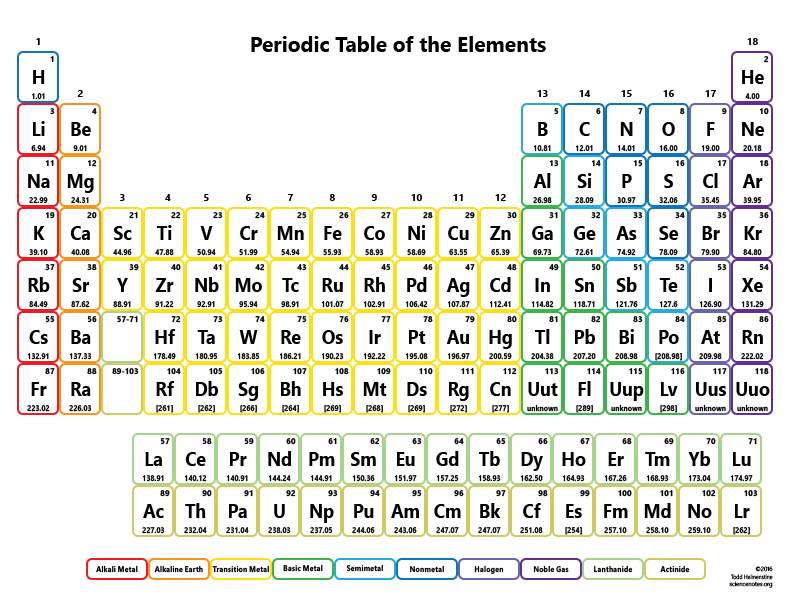
4/17/2024 0 Comments Periodic table molar mass of f Hence the Molar mass of FeCl3 is 162.195 g/mol. So, Molar mass of FeCl3 Molar mass of 1 Iron (Fe) atom + Molar mass of 3 Chlorine (Cl) atoms. Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. You can see that in FeCl3, there is 1 Iron atom and 3 Chlorine atoms. :max_bytes(150000):strip_icc()/PeriodicTable-58b5d9253df78cdcd8d03ebb.jpg)
Next, you have 3 oxygen atoms, so you would multiply the atomic mass of O by 3 to obtain an atomic weight of 48.00 g/mol. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. From the chemical formula, there are 2 iron atoms, so the atomic mass has to be multiplied by 2, which gives you a value of 111.7 g/mol. Find the molar mass of Fe (55.85 g/mol) and O (16.00 g/mol). 
In our case, our two elements are iron and oxygen. Let us calculate the molecular weight of some common compounds. The molar mass of Fe2O3 is 159.70 g/(mol) To find the molar mass of a compound you would use the periodic table to find the molar mass of each element separately. A column on the periodic table is known as a group or family. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. The periodic table has all sorts of cool information just based on its layout. One mole of 'something' contains 6.022 x 1023 entities. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (F (fluorine)) Count i Weight i. Then, lookup atomic weights for each element in periodic table: F: 18.9984032. A mole is the unit that measures the amount of a substance. First, compute the number of each atom in F (fluorine): F: 1. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
